FDA label requirements for dietary supplements
Defined by Congress in 1994 and regulated by the FDA, The Dietary Supplement Health and Education Act requires the following information on all nutraceutical packaging and labels:- Identity
- Net Quantity of Contents
- Supplement Facts
- Ingredient List
- Name and Address of the Business or Manufacturer, Packer or Distributor
Not only do these statements need to be visible on a dietary supplement label, but they must be placed in specific locations to remain compliant with FDA regulations. All statements must be located either on the front Principal Display or the Informational Panel.
The Principal Display Panel
The Principal Display Panel is the front-facing portion of the label and is the section most likely to be seen first by the consumer. The identity and net quantity statements must be placed on this section of the packaging:
1. Statement of Identity – The name of the nutraceutical product. If no common name is defined for the product, then this statement must appropriately describe the purpose of the supplement.
2. Net Quantity of Contents Statement – The amount of supplement that the container holds. This measure must be expressed in either numerical value, volume, or weight. If using volume or weight measurements, you must specify the quantity in both metric and imperial terms.
The Informational Panel
The Informational Panel is the portion of the label directly to the right of the Principal Display Panel. The supplement facts, ingredient list, and business contact information must be placed on this section of the packaging: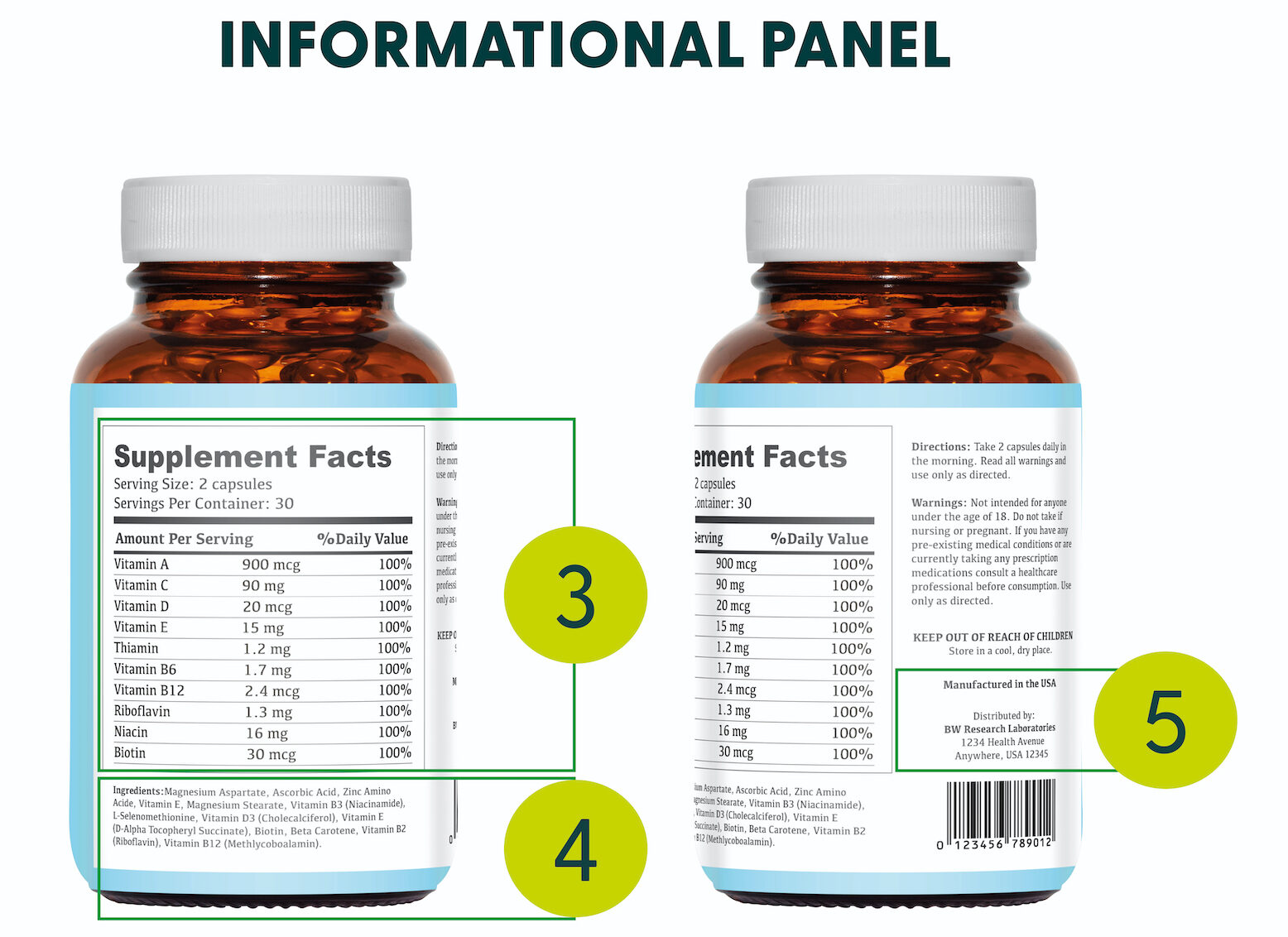
3. Supplement Facts Panel – The names and quantities of ingredients found in the supplemental product. These are listed along with the amount per serving and the percentage of the daily value of each. Additionally, the serving size and number of servings per container must be listed in this section.
4. Ingredient Statement – The compounds and substances used in manufacturing the contents of the dietary supplement. The ingredients must be listed in descending order based upon the weight of the ingredient itself.
5. Name and Address Statement – The name and location of either the manufacturer, packer, or distributor of the nutraceutical product. You must list the city or town, state, and zip code. You must also list the street address if it is not publicly available online or in a city directory.
Fonts and lettering requirements
To ensure readability for consumers, there are also supplement label requirements concerning text formatting for the FDA-required statements:
- Fonts must be easy to read.
- Letters must be at least 1/16th inch in height – using the lowercase “o” as a baseline.
- Letters cannot be greater than three times as high as they are wide – unless you file a petition with the FDA for an exemption.
- The copy must contrast with the background adequately to ensure ease of consumer readability.
Other FDA supplement label requirements
The FDA requires all supplement product labels to have the above information and follow legibility guidelines. But, if your product contains certain ingredients or uses particular marketing rhetoric, you’re not entirely done. There are additional stipulations in place that you may need to follow to ensure your supplement label is FDA-compliant:
Does your product contain iron?
The FDA requires specific warning statements for supplement products that contain iron. For such products, you must include the following warning statement on your label – copied verbatim:
“WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately.”
This statement must be placed on the information panel and be sectioned off from the rest of the copy by a hairline box. Additionally, this warning statement must be placed on any other labeling containing warnings and, if applicable, the product’s outer packaging.
Do you market your product with health claims?
Dietary supplements cannot be marketed as resources to treat, prevent or cure a disease or condition. The FDA defines products that make such claims as drugs, which pushes them into a different regulatory category.
However, nutraceutical brands can include health claims that their product increases overall well-being. For example, many supplements state on their packaging that they are a “high source” or “good source” of certain nutrients. To make a claim that your product is a “high source” of a specific nutrient, the product must contain at least 20 percent of the Daily Value of the nutrient. For a “good source” claim, the supplement must contain between 10-19 percent of the nutrient’s Daily Value.
If you do market your dietary supplement product as being designed to address a specific nutrient deficiency, you must include the following statement – copied verbatim:
“This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.”
Following FDA guidelines for nutraceutical labeling is crucial, as failure to comply could result in a large fine. After ensuring your supplement label is FDA-compliant, considering certain design and packaging elements is the next step in elevating your brand.
Read our latest blog post on nutraceutical packaging trends
DISCLAIMER: The information provided in this article does not, and is not intended to, constitute legal advice. All information, content, and materials provided are for informative purposes only and may not encompass or represent the most up-to-date legal or other information. To learn more about current nutraceutical label requirements and specific compliancy information, please visit the FDA’s website.


